biotechrabbit™ T4 DNA Ligase Rapid is an exceptionally pure, highly concentrated ligase for applications in which high enzyme concentrations are required. It is especially recommended for fast ligations.
T4 DNA Ligase Rapid is supplied with 5× Rapid Ligation Buffer containing PEG for fast 5–10 minutes ligation, ligation of low-concentration or blunt-end DNA.
T4 DNA Ligase catalyzes the formation of a phosphodiester bond between the terminal 5' phosphate and the 3' hydroxyl groups of duplex DNA or RNA. The enzyme efficiently joins blunt and cohesive ends and repairs single stranded nicks in duplex DNA, RNA or DNA–RNA hybrids.
Component | Composition |
T4 DNA Ligase Rapid | T4 DNA Ligase Rapid, 600 U/µl, in storage buffer containing 50% (v/v) glycerol. |
5× Rapid Ligation Buffer | Optimized buffer including PEG for rapid ligation. |
STORAGE | -20°C (until expiry date – see product label) |
Unit Definition
One unit of T4 DNA Ligase is defined as the amount of enzyme needed to ligate 50% of 100 ng of DNA fragments with cohesive ends in a 50 µl reaction mixture in 1× Ligase buffer incubated at 23°C for 30 minutes. Approximately 22 biotechrabbit cohesive end units are equivalent to one Weiss unit.
Quality Control
Protein Purity
Protein purity is confirmed by SDS polyacrylamide gel electrophoresis followed by Coomassie Brilliant Blue staining. Purity is >95%.
Exonuclease Activity
Linearized lambda/HindIII DNA fragments are incubated with the enzyme in a 50 µl reaction mixture for 4 h at 37°C. No DNA degradation observed.
Endonuclease/Nick Activity
Supercoiled plasmid DNA is incubated with the enzyme in a 50 µl reaction mixture for 4 h at 37°C. No conversion of covalently closed circular DNA to nicked DNA detected.
Contamination with E. coli DNA
Absence of E. coli genomic DNA is confirmed by qPCR using a sample of the enzyme and specific primers targeting the E. coli 16S rRNA gene. No contamination detected.
Fast ligation
- Thaw on ice and mix all reagents well.
- Keep all reagents and reactions on ice during setup.
- Single-insert ligations are optimal with an insert to vector ratio between 2 and 6.
- A ratio above 6:1 will promote the insertion of multiple fragments, while ratios below 2:1 will reduce ligation efficiency.
- For difficult ligations or if the DNA concentration is unknown, it may be necessary to vary ratios and run multiple ligations.
- Prepare the ligation reaction as shown below:
Component | Volume | Final concentration |
5× Rapid Ligation Buffer | 10 µl | 1× |
Vector DNA | Variable | 1–10 ng/µl |
Insert DNA | Variable | 1–10 ng/µl |
T4 DNA Ligase, Rapid (600 U/ µl) | 1 µl | 30 U/µl |
Nuclease free water | Variable |
|
Total volume | 20 µl |
|
- Mix and centrifuge briefly to collect the liquid in the bottom of the tube.
- Incubate at 25°C for 5–10 minutes.
- Heat inactivate the enzyme 65°C for 10 minutes or at 70°C for 5 minutes.
- For transformation, immediately purify or dilute DNA (see below).
Transformation into competent cells
PEG at a high concentration significantly reduces the transformation efficiency of electrocompetent cells. Before electrotransformation, PEG should be removed by one of the following alternative approaches:
- The preferable approach for efficient electroporation is to purify the ligation product using a DNA purification spin column (e.g. BR0700301 GenUP™ PCR Cleanup Kit), eluting in 50 µl of TE.
- Alternatively, dilute the ligation reaction with double-distilled water or TE at least twofold to reduce the PEG concentration. This approach is sufficient to prepare the ligation mixture for chemical transformation. For electrotransformation the first approach gives better results.
- The amount of DNA to be transformed should be in the range of 0.1–10 ng.
References
- Engler, M.J. and Richardson, C.C. (1982) P.D. Boyer (Eds.), The Enzymes, 5, pp. 3. San Diego: Academic Press.
1. Engler, M.J. and Richardson, C.C. (1982) P.D. Boyer (Eds.), The Enzymes, 5, pp. 3. San Diego: Academic Press.
You may also be interested in the following product(s)
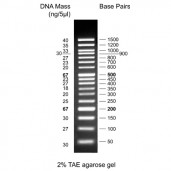
50 bp DNA Ladder, ready to use
Package Sizes |
UPstart™ Plus Taq Antibody, 1 mg/ml
Package Sizes |
RTS Linear Template Fab Kit
Package Sizes |

TriColor Protein Ladder (10-180 kDa)
Package Sizes |
A-Tailing Kit
Package Sizes |
